Article Text
Abstract
Background Many studies reported high prevalence of antiphospholipid antibodies (aPL) in patients with COVID-19 raising questions about its true prevalence and its clinical impact on the disease course.
Methods We conducted a meta-analysis and a systematic review to examine the prevalence of aPL and its clinical impact in patients with COVID-19.
Results 21 studies with a total of 1159 patients were included in our meta-analysis. Among patients hospitalised with COVID-19, the pooled prevalence rate of one or more aPL (IgM or IgG or IgA of anticardiolipin (aCL) or anti-ß2 glycoprotein (anti-ß2 GPI) or antiphosphatidylserine/prothrombin, or lupus anticoagulant (LA)) was 46.8% (95% CI 36.1% to 57.8%). The most frequent type of aPL found was LA, with pooled prevalence rate of 50.7% (95% CI 34.8% to 66.5%). Critically ill patients with COVID-19 had significantly higher prevalence of aCL (IgM or IgG) (28.8% vs 7.10%, p<0.0001) and anti-ß2 GPI (IgM or IgG) (12.0% vs 5.8%, p<0.0001) as compared with non-critically ill patients. However, there was no association between aPL positivity and mean levels of C reactive protein (mean difference was 32 (95% CI −15 to 79), p=0.18), D-dimer (mean difference was 34 (95% CI −194 to 273), p=0.77), mortality (1.46 (95% CI 0.29 to 7.29), p=0.65), invasive ventilation (1.22 (95% CI 0.51 to 2.91), p=0.65) and venous thromboembolism (1.38 (95% CI 0.57 to 3.37), p=0.48).
Conclusions aPLs were detected in nearly half of patients with COVID-19, and higher prevalence of aPL was found in severe disease. However, there was no association between aPL positivity and disease outcomes including thrombosis, invasive ventilation and mortality. However, further studies are required to identify the clinical and pathological role of aPL in COVID-19.
- COVID-19
- antibodies
- anticardiolipin
- antibodies
- antiphospholipid
Data availability statement
Data are available in a public, open access repository.
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Key messages
What is already known about this subject?
Antiphospholipid antibodies (aPLs) were frequently reported in patients with COVID-19. However, its true prevalence and its clinical impacts are unknown.
What does this study add?
We conducted the largest meta-analysis to date examining the prevalence and the clinical impact of aPL on the clinical features of patients with COVID-19. Our significant findings are: (1) nearly half of patients with COVID-19 were positive for one of the aPL. (2) Most frequently reported aPL was LA. (3) aPLs were significantly more frequently reported in critically ill patients, and (4) aPLs were not significantly associated with disease outcomes like venous thrombosis, invasive ventilation and mortality.
How might this impact on clinical practice or further developments?
The frequently reported aPL in patients with COVID-19, especially in critically ill patients, raises questions about its role in the pathogenesis of the disease.
Introduction
Hypercoagulability is one of the striking features of COVID-19. In a recent large study, risk of venous thromboembolism in patients with COVID-19 was 16%, while risk of arterial thrombosis was 11.1%.1 Others documented cerebral ischaemic infarcts in these subjects.2 In addition to macrothrombi, several autopsy studies of patients with COVID-19 revealed features of microangiopathy with microthrombi in various organs including, lung, kidney, heart, skin and prostate.3 4 Case fatality appears to be determined by formation of vascular thrombi in association with progressive severe endothelial injury in COVID-19 infected subjects.5 The clinical features related to vasculopathy and thromboembolism in patients with COVID-19 are wide ranging from asymptomatic with mild elevation of D-dimer to severe organ dysfunction due to macrothrombi and microthrombi.5 The pathogenesis of hypercoagulability in COVID-19 is not fully understood. Yet, SARS-CoV-2 mediated coagulopathy appears to have distinct features, such as normal to elevated fibrinogens, elevated D-Dimers, normal platelets and mild prolonged activated partial thromboplastin time.6 To identify the possible causes of macroangiopathy and microangiopathy in this disease, numerous studies evaluated the potential role of anticardiolipin antibodies (aPLs).2 7–26 In the current study, we conducted a meta-analysis and a systematic review to investigate if there is an association between COVID-19 and aPL.
Material and method
Aims and overview
The aims of this meta-analysis study are to describe the actual prevalence of aPL in patients with COVID-19 and to describe the potential clinical impact of positive aPL on the disease outcome.
Search strategy and study selection
The systematic review and meta-analysis were conducted adhering to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.27 We identified articles through a search of PubMed, MEDLINE, Embase, Web of Science and Google Scholar from 31 December 2019 to 15 October 2020. The following search terms were used: “COVID-19”, “SARS-COV-2”, “Antiphospholipid”, “Anticardiolipin”, “Lupus anticoagulant” and “Anti-B2 glycoprotein”. Studies reporting aPL in patients with COVID-19 were extracted. We also reviewed the references of each study to identify further related articles for analysis. There was no language restriction. The two investigators independently performed the search and determined the eligibility of studies according to the criteria mentioned below. Data extracted from the included studies using a data extraction form developed in MS Excel (online supplemental table 1). Selection results have been reported according to the PRISMA flow chart (figure 1).
Supplemental material
Searching and selection process.
Inclusion and exclusion criteria
All studies met the following criteria: (1) subjects were adults and diagnosed with COVID-19 based on RT-PCR or serum serological testing, (2) any of the following aPL tests performed: IgM or IgG of anticardiolipin (aCL) or anti-ß2 glycoprotein (anti-ß2 GPI) or lupus anticoagulant (LA) or antiphosphatidylserine/prothrombin (aPS/PT), (3) study sample was larger than 10 and (4) subjects had been randomly selected. We excluded articles with <10 subjects, case reports and paediatric population (<18 years old). In addition, we excluded articles with non-randomly selected population (eg, COVID-19 with cerebrovascular accidents or thromboembolism).
Antiphospholipid assay
aCL, anti-ß2 GPI and aPS/PT were detected in all studies using either ELISA or chemiluminescent immunoassay. The cut-off value for positive result was >20 U for aCL and anti-ß2 GPI and >30U for aPS/PT (except for study,26 the cut-off value for positive result was >15 U for aCL and >8U for anti-ß2 GPI). Seven studies9 13–15 18 19 22 did not mention the cut-off value for positive result. We considered positive titre according to the authors’ definition.
All studies mentioned the diagnostic method for detecting LA except two studies.21 24 The rest of the studies detected LA using dilute Russell’s viper venom time (dRVVT) screen, mixing and confirm assay. Not all studies reported details about heparin dose. Among the studies for which LA testing was conducted and details about heparin dose were reported, 71% subjects were on prophylactic dose, while the rest received therapeutic dose.
Statistical analysis
The statistical analysis was performed using Review Manager (V.5.4.1) provided by The Cochrane Collaboration. P value was two tailed, and the statistical significance set at ≤0.05. Weighted pooled prevalence and 95% CIs of aPL, aCL, LA and anti-ß2 GPI were calculated using a random-effects model. Weighted pooled prevalence of aPL, aCL, LA and anti-ß2 GPI was compared between intensive care unit (ICU) and non-ICU patients. All studies reported prevalence of aPL without a control group, so OR was not calculated. The association between aPL status and hospital outcomes (thrombosis, invasive ventilation and in-hospital mortality) was studied and expressed as OR with pertinent 95% CI. The association between aPL status and D-dimer and C reactive protein (CRP) was also examined and expressed as OR with pertinent 95% CIs. D-dimer and CRP were described using mean and SD. Unit discordance for variables was resolved by converting all units to a standard measurement for that variable. A random-effects model was used to combine data. We assessed the heterogeneity in this meta-analysis by using the I2 statistical test. Assessment for publication bias was not done as less than 10 studies were included in each analysis so the power of the tests is too low to distinguish chance from real asymmetry.
Results
Study selection
After searching database, we identified a total of 47 studies. After scanning the title/abstract, case reports, review articles, editorials, letters and comments, and duplicate studies were excluded. Further selection yielded to 32 potentially relevant studies that we accessed for further eligibility. Of these, 11 studies were excluded for the following reasons: paediatric population <18 years old, the study population was not randomly selected, and study sample was <10 subjects. Thus, 21 articles reflecting 1159 patients were included for analysis (online supplemental table 1). The searching and selection process is summarised in (figure 1).
Study characteristics
The selected articles2 7–26 and extracted data are included in (online supplemental table 1). All the studies were either cross-sectional or retrospective observational studies. The frequency of aPL was reported in all studies. aPLs were reported in hospitalised patients, either critically ill patients or non-critically ill patients or both: 10 studies conducted on critically ill patients,2 7 9 10 12 15 18 19 24 25 1 study in non-critically ill patients21 and 10 studies in both.8 11 13 14 20 22 23 26 28 29 We found nine studies were comparing patients with positive and negative aPL.7 9 10 13 18 22 24 28 29 SARS-CoV-2 infection was confirmed in all studies, and the predominate reported confirmation method was RT-PCR test. However, few studies,9 11 15 19 20 25 did not specify which method was used to confirm SARS-CoV-2 infection. Information about when the laboratory testing for cardiolipin antibodies was performed as well as information about in-hospital treatment with antiviral, corticosteroids or heparin during hospitalisation was not available in most of the studies.
Prevalence rates of aPL in patients with COVID-19
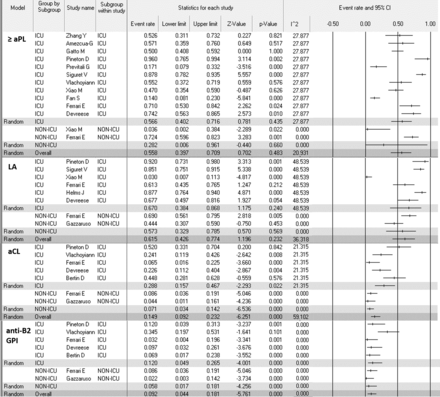
In this analysis, we included the prevalence of antibodies for all studies. A total of 1159 patients (from 21 studies) hospitalised with COVID-19 and had one of the aPLs reported were included in this analysis. The pooled prevalence rate of one or more aPL (IgM or IgG of aCL or anti-ß2 GPI or aPS/PT, or LA) was 46.8% (95% CI 36.1% to 57.8%) (table 1). The most frequent type of aPL found was LA, with pooled prevalence rate of 50.7% (95% CI 34.8% to 66.5%) (table 1). The pooled prevalence rate of aCL (IgM or IgG) and anti-ß2 GPI (IgM or IgG) were 13.9% (95% CI 7.5% to 24.1%) and 6.7% (95% CI 3.5% to 12.5%), respectively (table 1). Four studies9 17 20 22 reported double and triple aPL positivity. Pooled prevalence was 14.3% (95% CI 4.0% to 40.3%) for double aPL positivity and 6.1% (95% CI 2.8% to 12.7%) for triple aPL positivity. I2 test did not show significant heterogenicity between studies (table 1).
Weighted pooled prevalence of LA, ACL, anti-ß2 GPI and any APL in patients hospitalised with COVID-19
Repeated antiphospholipid assay
Two studies retested aPL in few patients with COVID-19 at later time point during hospitalisation. One study25 found that 9 out of 10 LA-positive patients were retested negative at later time. The other study28 retested aPL in six patients at multiple time-points and found that aPL levels, in general, reached a peak, which then declined over 3–4 week follow-up periods. These results suggest that aPL may be transiently elevated in patients with COVID-19.
Prevalence of aPL in ICU versus non-ICU patients with COVID-19
This meta-analysis found that critically ill patients with COVID-19 had significantly higher prevalence of aCL (IgM or IgG) (28.8% vs 7.10%, p<0.0001) and anti-ß2 GPI (IgM or IgG) (12.0% vs 5.8%, p<0.0001) as compared with non-critically ill patients (figure 2). However, there was no statistically significant difference in prevalence of LA or aPL in general between critically and non-critically ill patients with COVID-19 (figure 2). I2 test did not show significant heterogenicity between studies (figure 2).
Forest plot on the prevalence of aPL in ICU versus non-ICU patients with COVID-19. aCL, anticardiolipin; anti-ß2 GPI, anti-ß2 glycoprotein; aPL, anticardiolipin antibodies; ICU, intensive care unit; LA, lupus anticoagulant.
Outcomes of COVID-19 related hospitalisation in positive versus negative aPL
Eight studies2 8 11 14 16 20 22 25 examined the association of aPL positivity and hospital outcomes. Importantly, pooling data from these studies showed no association between aPL positivity and mean levels of CRP (mean difference was 32 (95% CI −15 to 79), p=0.18) and D-dimer (mean difference was 34 (95% CI −194 to 273), p=0.77) (figure 3). I2 test did show significant heterogenicity between studies in CRP analysis (I2=75%, p=0.008) but not in D-dimer analysis (I2=0.0%, p=0.57).
Forest plot on the mean difference of CRP (A) and D-dimer (B) in aPL-positive patients with COVID-19 compared with aPL-negative patients with COVID-19. aPL, anticardiolipin antibodies; CRP, C reactive protein.
Similarly, there was no association between aPL positivity and mortality (1.46 (95% CI 0.29 to 7.29), p=0.65), invasive ventilation (1.22 (95% CI 0.51 to 2.91), p=0.65) and venous thromboembolism (1.38 (95% CI 0.57 to 3.37), p=0.48) (figures 4 and 5). There was significant heterogeneity between studies in mortality (I2=74%, p=0.004) and venous thromboembolism (I2=68%,p=0.003) but no heterogeneity in invasive ventilation analysis (I2=19%, p=0.029). The heterogeneity observed across studies can be explained by methodological differences like different type of aPL tested, different methods of testing and different positivity cut-off.
Forest plot on the odds of mortality (A) and invasive ventilation (B) in aPL-positive patients with COVID-19 compared with aPL-negative patients with COVID-19. aPL, anticardiolipin antibodies.
Forest plot on the odds of venous thromboembolism in aPL-positive patients with COVID-19 compared with aPL-negative patients with COVID-19. aPL, anticardiolipin antibodies.
Discussion
In this study, we conducted the largest meta-analysis to date examining the prevalence of aPL in COVID-19 and the association between aPL and disease severity. Our significant findings are: (1) nearly half of patients with COVID-19 were positive for one of the aPL; (2) most frequently reported aPL was LA; (3) aCL and anti-ß2 GPI were significantly more frequently reported in critically ill patients; and (4) aPLs were not significantly associated with disease severity.
The association between aPL and viral infections is widely acknowledged.30 We compared findings of this study with previous studies that examined the prevalence of aPL in other viral infections.30 31 We find that the prevalence of aCL was much lower, and the prevalence of LA was strikingly higher in our analysis related to SARS-CoV-2 compared with the ones previously reported in other viral infections.31 For instance, a previous meta-analysis31 reported very high prevalence of aCL in HIV (56%), Epstein-Barr virus (EBV) (50%) and hepatitis C virus (HCV) (21%) compared with the aCL prevalence in our analysis (13.9% (95% CI 7.5% to 24.1%)). In addition, the same study reported low prevalence of LA in HIV (2%), HCV (<1%) and hepatitis B virus (HBV) (1%) compared with the LA prevalence in our analysis (50.7% (95% CI 34.8% to 66.5%)). In other words, it looks like the predominant pattern of aPL in viral infections has shifted from aCL in HIV, HCV and EBV to LA in SARS-CoV-2. Among antiphospholipid antibodies, it is well known that presence of LA is strongly associated with thrombosis compared with aCL and anti-ß2 GPI.32
However, the high prevalence of LA in this analysis must be interpreted carefully as LA testing results, in contrast to aCL and anti-ß2 GPI testing, can be affected by heparin administration and cause false positive results.33 34 Current guidelines33 34 recommend caution when interpreting LA testing in patients receiving heparin and recommend blood to be drawn for LA testing after 12 hours since the last dose of heparin. Unfortunately, this was not considered when interpreting LA results in all of the studies included in this meta-analysis except in one study.22 Thus, heparin administration is a potential bias in our analysis; however, the three-step (dRVVT) screen test that has been used in the studies of this meta-analysis can neutralise any heparin effect, especially in prophylactic dose.35 Seventy-one per cent of this analysis subjects received prophylactic dose, while the rest received therapeutic dose, and according to British Committee for Standards in Haematology guideline,34 prophylactic doses of heparin should have less effect on LA testing results. Therefore, the fact that dRVVT screening test was used in this analysis subjects and most of them received prophylactic dose of heparin may reduce the risk of bias related to heparin administration. In summary, despite the potential bias of LA testing by heparin administration, LA may be truly elevated in SARS-CoV-2 compared with other viral infections. This finding requires further investigations to examine the true prevalence and role of LA in SARS-CoV-2 infection.
One of our significant findings of this study is that severe COVID-19 disease requiring ICU care is associated with higher prevalence of aCL and anti-ß2 GPI. In contrast, severe disease was not found to be associated with the prevalence of LA. However, this finding might be biased by heparin administration that interfere with LA testing as explained before. High prevalence of aPL in critically ill patients has been reported previously.36 This can be partially explained by the extensive inflammation, cellular damage and apoptosis in critically ill patients that can induce aPL production.37 38 aPL are antibodies targeting mainly phospholipid-binding proteins, such as β2-GPI and prothrombin, that expressed on cell membrane at high density.38 It was hypothesised that the damaged apoptotic cell surfaces expose these cellular components to the immune system, predisposing an individual to develop aPL.38 Although aPL is associated with critical illness, this analysis as well as previous studies36 39 did not find a significant association between aPL and disease outcomes like invasive ventilation and mortality. These findings suggest that aPL may be markers for disease severity or tissue injury but if aPL contribute to tissue damage and disease severity is questionable and require further investigations.
Surprisingly, the presence of aPL was not significantly associated with elevated D-dimer or thrombosis. This may be explained by the low titres or transient elevation of aPL in COVID-19 as compared with patients with antiphospholipid syndrome.16 25 Another interesting explanation is that aPL found in viral infections or healthy aPL carriers might be different and less pathogenic than the ones found in antiphospholipid syndrome. A recent study showed that healthy aPL carriers had higher anti-β2 GPI-D4/5 but lower anti-β2 GPI-D1 compared with patients with antiphospholipid syndrome.40 Anti-β2 GPI-D1 subclass is associated with higher risk of thrombosis compared with anti-β2 GPI-D4/5.41 In addition, it was found that purified anti-β2GPI-D5, unlike anti-β2GPI-D1, did not recognise cardiolipin-bound β2 glycoprotein I while being able to interact with β2 glycoprotein.42 This might explain the higher prevalence of anti-ß2 GPI compared with aCL reported in some of the studies included in this analysis.15 16 18 20 To our knowledge, none of the studies subclassified IgG aPL in patients with COVID-19; therefore, subclassification of IgG aPL in COVID-19 is needed.
Although we found that the presence of aPL was not significantly associated with thrombosis, a direct relationship between LA alone and thrombosis has not been studied. Also, none of the studies examined the relationship between combined positivity for aPL and thrombosis knowing that combined positivity for aPL carries higher risk of thrombosis.32 Additionally, heparin was administered to most of the subjects included in this meta-analysis at either prophylactic or therapeutic dose. Heparin administration may have decreased macrothrombosis and confounded the association between aPL and thrombosis in patients with COVID-19. In contrast to macrothrombosis, the direct measurement of microangiopathy and microthrombi is difficult in living organisms. Microthrombi in the small lung vasculatures is a common microscopic finding, occurring in 80%–100% of lungs examined at autopsy.4 Thus, despite the lack of association between the presence of aPL and macrothrombosis, the presence of aPL may have contributed to the widespread microvascular thrombosis seen in patients with COVID-19.3 4
In summary, aPL were frequent in patients with COVID-19, and severe disease is associated with higher prevalence of aPL. Although our analysis showed that the presence of aPL was not associated with thrombosis, invasive ventilation and mortality, these findings still cannot rule out any contribution of aPL in the pathogenesis of COVID-19.
Conclusion
Based on this meta-analysis, we concluded that the prevalence of aPL is high in patients with COVID-19, and LA is the most frequent aPL reported. The prevalence of aPL was higher in ICU compared with non-ICU patients. Our analysis found no correlation between aPL positivity and disease outcomes such as thrombosis, invasive ventilation and mortality. These findings may suggest that aPL are just markers for disease severity rather than being pathogenic. However, further investigations are still required to identify the contribution of these markers in the pathogenesis of COVID-19.
Limitation
Due to meta-analysis nature, our results have several limitations: (1) as discussed previously, thrombotic events reported in some studies without mentioning if the prophylactic or therapeutic heparin were administered. (2) Heparin administration and elevated CRP may impact the LA testing results. (3) Most studies did not mention when aPL was measured during hospitalisation. (5) Not all studies excluded patients with previous history of autoimmune diseases or thrombophilia. (6) Most studies used the value >20 U as a cut-off value for positive result; however, several others9 13–15 18 19 22 did not specify the exact cut-off value. This cut-off value is apparently low, and likely borderline results were considered positive. Low aPL titres might have low prognostic values, whereas the higher aPL titre (>40 U) is well known to carry higher risk for thrombosis and is required for the diagnosis of antiphospholipid syndrome.
Data availability statement
Data are available in a public, open access repository.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Contributors MT: collecting data, analysis and writing manuscript. LS: collecting data, analysis, writing manuscript and correction.
Funding The work was supported by a grant by NHLBI (R01HL150474) to LS.
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.