Abstract
Objectives
According to the recent epidemiological studies, patients with diabetes mellitus (DM) may be at higher risk of hospitalization due to COVID-19. Regarding the important role of nutrition on the immunity, the present review article aimed to outline nutritional support of DM during the outbreak of COVID-19 with a mechanistic insight.
Methods
Searches were performed in PubMed/MEDLINE, ScienceDirect, Scopus, and Google Scholar databases from 2000 until December 2020 using the following keywords. All relevant clinical and experimental studies published in English were included.
Results
Evidences revealed that hyperglycemia is a significant predictor of some viral infections including COVID-19 which can exacerbate the complications of DM. According to the literature review, adequate intake of dietary protein, fiber, essential fatty acids and some micronutrients especially vitamins D, C, B12, folate, zinc and selenium has beneficial effects on the prevention and treatment of COVID-19 in diabetic patients through modulation of innate and adaptive immune responses or direct effects on virus enzymes or the rate of cell entrance.
Conclusions
It is well understood that malnutrition may increase susceptibility to viral infections and disease progression. Therefore, considering nutritional status of diabetic patients and reasonable supplementation of the above mentioned nutrients can ameliorate the symptoms of COVID-19 in DM. However, further well-designed clinical trials are needed to determine their therapeutic dose.
Similar content being viewed by others
Introduction
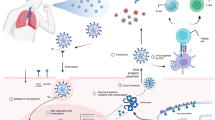
Nowadays, the novel coronavirus (COVID-19) is the main health care problem around the world [1]. According to the latest statistics of World Health Organization (WHO), there are more than 86 million confirmed COVID-19 cases and more than 1.5 million deaths around the world [2]. Clinical manifestations of COVID-19 range from mild symptoms to critical illness and death [3, 4]. Frequently reported symptoms of infected patients include fever (77–98 %), cough (46–82 %), myalgia or fatigue (11–52 %), and difficulty of breath (3–31 %) at the onset of illness, which can progress to sever lung injury and severe respiratory distress syndrome [3, 5,6,7]. It was reported that the virus entry in to the alveolar epithelial cells may be through the angiotensin-converting enzyme-2 (ACE2) receptor which can cause severe lung inflammation and acute respiratory distress syndrome (ARDS) [8,9,10].
Available researches indicated that older adults and people with underlying diseases including diabetes mellitus (DM), cardiovascular disease, hypertension, lung disease and cancer are at higher risk and more prone to the infection [11,12,13,14].
It was reported that patients with DM have had higher morbidity and mortality rates of COVID-19. According to a recent systematic review, the incidence rate of COVID-19 in diabetic patients was 14 % [15]. Docherty et al., reported that among 20,133 hospitalized patients with severe COVID-19 in the UK, the prevalence of DM was 21 % [16]. Moreover, Riddle et al., reported that among 44,672 patients in China with confirmed COVID-19, the mortality rate of diabetics was 7.3 %, which was more than three times that of the overall population [17]. Another study in the UK reported that the mortality risk of diabetics with COVID-19 can be up to 2–3 times greater than non-diabetic patients [18]. However, Fadini et al., indicated that DM may not increase the risk of COVID-19 incidence but can worsen its outcome and severity of the infection [19]. According to the epidemiological studies, DM increases the risk of hospitalization, critical care requirement and also mortality rate due to COVID-19 [20,21,22].
Therefore, it seems that diabetic patients need special attention during the pandemic of COVID-19.
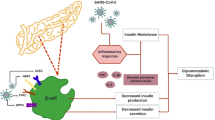
Diabetes is a chronic inflammatory condition that can affect immune response to pathogens [22]. Hyperglycemia promotes secretion of pro-inflammatory cytokines and glycosylation end products (AGEs) which makes patients vulnerable to the infections [23, 24]. In addition, over production of free radicals after viral infection can exacerbate the oxidative stress in diabetics and leads to the intrapulmonary oxidative injury and lung inflammation [19].
It is well understood that malnutrition may affect the consequences of viral infections and also its pathogenicity [23,24,25,26,27,28]. Researchers indicate that host nutritional status not only affects the immune function of the body, but also may have some direct effects on the viral genome and its virulence [29]. Therefore, considering nutritional status of people, especially most vulnerable patients like diabetics during the outbreak of novel coronavirus is very important. In this regard, the present review article aimed to outline nutritional support for the prevention and treatment of COVID-19 in diabetic patients, with a focus on nutrients’ mechanisms of action.
Methods
Searches were performed in PubMed/MEDLINE, ScienceDirect, Scopus, and Google Scholar databases from 2000 until December 2020 using the following keywords based on MeSH terms. Generally, a topic-centric search was conducted to write each section. We included all relevant clinical and experimental studies which were the closest to our keywords and published in English. The search terms included “immunity” OR " viral infection” OR " COVID-19” AND “diabetes” AND/OR “nutrition” OR “antioxidant”; “vitamin D”; “vitamin A”; “vitamin C”; “folic acid”; “zinc”; “selenium”; “macronutrient “. These search items were used for the relevant sections of the review.
As the number of studies on nutritional support of COVID-19 and diabetes mellitus is very limited, we included all related antiviral nutrients along with recent reports regarding their effects on COVID-19 and diabetes mellitus. Data were extracted from the included manuscripts by one author (SM). Two other authors checked the accuracy of the data extracted (VEA, MSA). Some of the related studies are mentioned in the Table 1.
Results and discussion
Macronutrients
Carbohydrates
It was reported that hyperglycemia is a significant predictor of some viral infections including H1N1 [39], SARS-COV [40] and MERS-COV [41]. Moreover, it seems that there is a bilateral association between blood sugar and infection. A recent study demonstrated increased secretion of glucocorticoids and catecholamines, insulin resistance and increased blood glucose levels after infection with COVID-19 which can exacerbate diabetic complications [42]. Therefore, it is important to pay attention to the glycemic control of viral-infected patients especially those with DM.
Diabetic patients with mild to moderate COVID-19 symptoms need a balanced ratio of carbohydrates intake (45–55 % of calories) mostly as complex carbohydrates and low simple sugars for better glycemic control. In addition, adequate intake of functional fibers (prebiotics such as beta-glucan and fructo-oligosaccharides) from dietary sources (e.g., oat, wheat, banana, onions, garlic, and tomato) can be helpful through direct effect on gut microbiota and also immune defense system [43,44,45].
Hospitalized diabetic patients with severe COVID-19 symptoms may need supportive nutrition. Enteral nutrition (EN) is usually well tolerated by the patients and is preferred over parenteral nutrition (PN). Nasogastric or orogastric EN should be initiated within 48 h of admission to the intensive care unit (ICU) [46,47,48]. The amount of carbohydrate in enteral feeding formula depends on patient’s condition (e.g., glycemic control, ventilator dependency) and is usually 30–50 % of non-protein calorie [49]. Generally, it was recommended that glucose should not exceed 5 mg/kg/min [46, 47].
Results of some experimental studies [43, 50] showed that significant restriction of carbohydrates can exacerbate viral outcomes because the immune cells like CD4 + and CD8 + T cells, involved in pathogen clearance, supply most of their energy from glucose and anaerobic glycolysis [51].
Protein
Some evidences support the beneficial effects of high protein diets on glycemic control, insulin resistance and maintenance of lean body mass in diabetic people [52]. The amount of protein intake can also directly impact the immune response. It was reported that mice feeding with a low-protein diet, followed by the infection with influenza virus was accompanied by decreased number and function of CD8+ T cells as well as NK cells, higher viral titers and increased mortality of animals [51, 53]. Therefore, appropriate protein intake of 1.2 to 1.5 g/kg ideal body weight (20–30 % of calories) with at least 50 % from high biological value (HBV) proteins is recommended for almost all people including type 2 diabetic adults and older people to maintain their respiratory muscle strength and support immune function during infection with COVID-19 [54, 55].
In turns of EN for critically ill patients with COVID-19, the American Society for Parenteral and Enteral Nutrition (ASPEN) recommended the protein supply of about 1.2–2.0 g/kg/day for non-obese patients (based on actual body weight) and 2-2.5 g/kg/day for class I- III obese patients (based on ideal body weight) [46,47,48]. When EN cannot be continued, PN should be prescribed with per case decision making [56].
Fatty acids
It was reported that the type of dietary fatty acid is very important in the inflammatory and immunomodulatory responses of host to a microbial agent [57, 58]. Adequate intake of omega-3 fatty acids may protect host against viral and bacterial infections [59]. However, there are some controversies regarding the efficacy of omega-3 supplementation in different bacterial and viral infections [60,61,62,63]. Patients with DM are more exposed to oxidative stress. Therefore, it is important to consider the quantity and also quality of dietary fatty acids especially during infections and inflammatory condition [64, 65].
COVID-19 can cause severe lung injury and ARDS [66]. In this condition, increasing dietary fat intake helps to decrease CO2 production as well as ventilator dependency. According to the dietary recommendations for ARDS and pneumonia, an enteral feeding with 30–45 % of calories from fat maybe helpful [67,68,69]. The amount of fat in EN formula depends on patient’s condition (e.g., serum triglyceride, ventilator dependency) and is usually 50–70 % of non-protein calorie [49].
However, regarding the quality of fatty acids, results of two recent systematic reviews showed that there are some controversies toward the benefits of long- chain omega3 fatty acids supplementation on the length of mechanical ventilation and mortality rate in adult patients with ARDS [69, 70]. Overall, adequate intake of essential fatty acids (EFAs) and appropriate proportion of poly unsaturated fatty acids (PUFAs) and mono unsaturated fatty acids (MUFAs) intake can be effective in patients with COVID-19 pneumonia and ARDS symptoms [48, 68, 71]. According to the recommendation of ASPEN for enteral feeding, 500 mg/day eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) might be added to the parenteral feeding requirements of 0.1 to 0.2 g/kg/d [46]. Moreover, the use of omega 6 soy-based intravenous lipid emulsion should be restricted [47].
Micronutrients
Vitamin B12 and B9
Vitamin B12 (VitB12) and vitamin B9 (folate/ folic acid) both act as a human immunity modulator. They can stimulate the T-lymphocytes production, restores the ratio of CD4/CD8 and improve beta-cell function by lowering oxidative stress [71,72,73,74,75,76]. Low serum levels of folate and B12 in type 2 diabetic patients is correlated with lower circulating level of glutathione and total antioxidant status [77]. Moreover, vitamin B12 supplementation in deficient subjects increased percentage of CD3 and CD7 cells and restored the function of lymphocytes and NK cells [78].
It should be noticed that diabetic patients are more prone to folate and B12 deficiency because of the established drug-nutrient interactions, especially those treated with metformin [79,80,81].
There is insufficient evidence about the effectiveness of B vitamins in the treatment of COVID-19 and it is not mentioned in the WHO Interim Guidance for COVID-19 management [82]. However, there are some promising reports about benefits of folate and VitB12 regarding the novel coronavirus (SARS-CoV-2).
Recently, Narayanan and Nair suggest that VitB12 can inhibit the RNA-dependent-RNA polymerase activity of the nsp12 molecule from SARS-CoV-2 virus and therefore decrease its pathogenicity and the severity of infection [31]. Two recent studies reported that supplementation with VitB12 may increase the synthesis of melatonin in body which has anti-inflammatory and anti-oxidative effects and may be effective in the treatment of COVID-19 [83, 84].
It was reported that SARS-CoV-2 may interfere with vitamin B12 metabolism and cause clinical manifestation of cobalamin deficiency by impairing the intestinal microbial proliferation. Therefore, it has been suggested that treatment with vitamin B12, especially as methyl cobalamin, can ameliorate symptoms of COVID-19. Accordingly, supplementation with high doses of VitB12 (1000–2000 µg/day) during the existing deficiency, can be effective [85].
Moreover, there are some recent promising reports of the effectiveness of folic acid on COVID-19. It was shown that folate can inactivates protease 3CLpro, a protein that all the coronaviruses need to replicate, so can reduce the replication of this virus [86, 87]. Two other studies reported that folate can inactivate the furin endoprotease enzyme of SARS-CoV-2 which is crucial for the virus entrance into the host cell [30, 88].
Vitamin D
Over the past decade, vitamin D (VitD) has attracted substantial interest due to its extra-skeletal functions including immuno-modulatory and anti-inflammatory effects [89,90,91]. Vitamin D has also some metabolic effects like stimulating insulin secretion and reducing peripheral insulin resistance through vitamin D receptors in the muscles and liver [92,93,94].
Regarding the immunomodulatory effect of VitD, results of some review articles showed that low circulating level of VitD is related to increased risk of viral respiratory infections [95,96,97] and VitD supplementation may has protective effects on the respiratory tract infections like influenza virus [98, 99].
In the case of current COVID-19 pandemic, it was suggested that vitamin D- deficient subjects may be more vulnerable to the infection [100] and there is a significant reverse association between serum 25(OH) D level and severity of COVID-19 disease [101]. Moreover, the outbreak of novel coronavirus occurred in winter, the time when 25-hydroxyvitamin D (25(OH) D) concentrations are lowest. Generally, the mortality rate of COVID-19 increases with age and comorbidities like diabetes which are associated with lower VitD concentration [33, 102].
Results of a recent retrospective study indicated that insufficient serum level of VitD was one of the main fatality risk factors related to COVID-19 and VitD insufficient cases were approximately 12.55 times more likely to die after the infection. Moreover, the combination of comorbidities with VitD insufficiency were associated with increasing odds of death [103].
It is established that VitD modulate both innate and adaptive immune responses, B-lymphocytes, monocytes, macrophages, dendritic cells (DCs) and T-cells. Vitamin D can suppress the pro-inflammatory Th1 and Th17 cells and also enhance the regulatory T cells [104]. Along with these immunomodulatory effects, the protective effect of VitD against COVID-19 is also related to the suppression of cytokine response and reduced risk of ARDS [105]. Moreover, VitD supplementation seems to inhibit ACE2, one of the main identified host cell receptors of COVID-19 and consequently helps to attenuate the lung infection [106].
As a preventive strategy, it is recommended to maintain serum level of VitD at sufficient level (> 30 ng/mL) and the goal should be to raise VitD concentrations above 40–60 ng/mL [107]. Overall, it was reported that VitD concentration of 38 ng/mL was appropriate to decrease pneumonia. However, to achieve the above 25(OH) D concentration range, supplementation with 2000–4000 IU/day was recommended especially for people at higher risk of COVID-19 including diabetic patients [32]. However, those health benefits of vitamin D should not be exaggerated and people should skip from toxic overdoses supplementation.
Vitamin C
Vitamin C (VitC) is a famous antioxidant and also has immune-modulating effects through T-cells transformation, interferon production and phagocytic function [108].
It was reported that patients with severe respiratory infections have lower plasma VitC concentrations so that treatment with VitC restores their plasma VitC levels and ameliorates severity of respiratory symptoms [109]. Hemila et al. demonstrated that the high-dose intravenous VitC infusion (200 mg/kg body weight/day) decreased the duration of hospitalization in severe sepsis and septic shock, along with a significant reduction in the mortality rate [110]. These findings were also reported in patients infected by influenza virus [111, 112].
There are some evidences that vitamin C deficiency is more prevalent in patients with diabetes mellitus compared to the non-diabetic individuals. DM is usually accompanied by increased production of reactive oxygen species (ROS) and decreased level of antioxidant enzymes like superoxide dismutase (SOD) [113, 114]. Numerous studies reported the protective effects of VitC against oxidative damage of diabetes [115, 116].
Nowadays during the pandemic of COVID-19, it has been reported that the markers of inflammation and oxidative stress increase in infected patients and large dose of antioxidants like VitC may be applicable to COVID-19 [117]. A most recent study by Hiedra et al., reported that short term intravenous vitamin C treatment in SARS-CoV-2 positive patients (n = 17) decreased inflammatory markers. Moreover, low mortality rate and the need for mechanical ventilation in this study may be related to VitC treatment. Interestingly, 47 % of those infected patients have diabetes mellitus [34].
To our knowledge, a few clinical trials has recently designed to assess the effect of vitamin C treatment in patients with COVID-19. Their results and also further randomized controlled trials in severe COVID-19 patients especially with the comorbidity of DM could better clarify the possible protective role of vitamin C.
Selenium
Selenium (Se) plays a functional role in redox hemostasis, hormone metabolism and protection against oxidative stress and inflammation [118]. Low serum level of selenium is related to poor immune function and increased risk of mortality and its deficiency impairs both the innate and acquired immunity [119, 120]. Moreover, some epidemiological studies reported that a high level of serum selenium reduce the prevalence of diabetes and also its chronic complications [121, 122].
Regarding the immunomodulatory effects of Se, it is well documented that selenium deficiency is associated with higher susceptibility to viral infections and their pathogenesis therefore, it seems that Se supplementation may be applicable for COVID-19 [123]. A most recent epidemiological study by Zhang et al., demonstrated a positive correlation between selenium status (based on hair selenium concentration) and the recovery rate of COVID-19 [35].
In another study Moghaddam et al., reported that high serum level of selenium in COVID-19 patients was related to increasing survival rate [36]. It seems that Se inhibits the entrance of different viruses into the cells and prevents their infectivity. Therefore, it may be useful against the pandemic of COVID-19 [124]. Since diabetic patients are susceptible to low anti-oxidative level, Se supplementation might be a helpful choice for the prevention or treatment of diabetic patients infected by novel coronavirus.
Zinc
Zinc (Zn) has a lot of functions in humans’ body as it is a cofactor for more than 300 enzymes. [125]. Besides, the antioxidant and anti-inflammatory effects of Zn, this element has vital roles in both innate and adaptive immune cells and also is essential for activation and secretion of insulin [126,127,128]. There are evidences of its protective effects against DM [129, 130].
Since zinc depletion deteriorates antiviral immunity, it is hypothesized that Zn deficiency may increase host susceptibility to COVID–19 [131].
Despite the lack of clinical data, some indications suggest that normal zinc status may be beneficial in COVID‑19. According to the in vitro studies, zinc has antiviral activity through inhibition of a number of RNA polymerases [37]. Velthuis et al. indicated that the increased level of intracellular zinc can effectively inhibit the replication of SARS–CoV in cell culture [38]. It was also reported that papain–like protein 2, a viral protease of SARS–CoV that is fundamental for its virulence, is potently inhibited by zinc [132]. This antiviral actions of zinc against SARS–CoV may also apply to SARS–CoV2, since their genome have some similarity. Recently, Shittu et al., demonstrated the improving efficacy of Chloroquine and Hydroxychloroquine against SARS-CoV-2 during combination with zinc supplementation [133]. Zinc may also have a modulatory effect on ACE2 activity, as the main receptor of SARS‑CoV‑2. Speth et al., showed that zinc exposure (100 µM) reduced recombinant human ACE‑2 activity in rat lungs [134].
Generally, according to the indirect evidences, Zn supplementation has a potential to decrease the incidence and also severity of SARS-CoV-2 infection, especially in groups with high risk of zinc deficiency including patients with DM, children and older adults [135].
Conclusions
Nutritional deficiencies of protein, essential fatty acids and some micronutrients are associated with decreased immune function and increased susceptibility to viral infections including the novel coronavirus which can affect severity of infection especially in vulnerable patients like diabetics. Therefore, considering nutritional status of patients and appropriate macronutrient and adequate micronutrients intake is very important in the management of COVID-19.
There are some promising reports of the effectiveness of adequate protein, essential fatty acids and some micronutrients especially vitamins D, C, B12, folate and zinc and selenium on the prevention and treatment COVID-19. According to some recent researches reasonable supplementation of those nutrients can ameliorate the symptoms of COVID-19 through modulation of innate and adaptive immune responses and also direct effects on virus enzymes or its entrance to the host cells. However, further well-designed clinical trials are needed to determine their treatment dose.
Data availability
No datasets were generated or analyzed during the current study.
Abbreviations
- 25(OH) D:
-
25-hydroxyvitamin D
- 3CLpro:
-
3 C-Like protease
- ACE2:
-
Angiotensin-converting enzyme-2
- AGEs:
-
Glycosylation end products
- ARDS:
-
Acute respiratory distress syndrome
- ASPEN:
-
American Society for Parenteral and Enteral Nutrition
- CD4+:
-
cluster of differentiation 4+
- CD8+:
-
cluster of differentiation 8+
- DCs:
-
Dendritic cells
- DM:
-
Diabetes mellitus
- DHA:
-
Docosahexaenoic acid
- EN:
-
Enteral nutrition
- EPA:
-
Eicosapentaenoic acid
- H1N1:
-
a subtype of Influenza A virus
- HBV:
-
High biological value
- MERS:
-
Middle East respiratory syndrome
- NK cells:
-
Natural killer cells
- nsp12:
-
non-structural proteins 12
- PN:
-
Parenteral nutrition
- RdRP:
-
RNA-dependent-RNA polymerase
- ROS:
-
Reactive oxygen species
- SARS-COV:
-
Severe acute respiratory syndrome coronavirus
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus 2
- Se:
-
Selenium
- SOD:
-
Superoxide dismutase
- Th1:
-
T Helper Cell Type 1
- Th17:
-
T Helper Cell Type 1
- VitB12:
-
Vitamin B12
- VitC:
-
Vitamin C
- VitD:
-
vitamin D
- WHC:
-
World Health Organization
- Zn:
-
Zinc
References
Palacios Cruz M, Santos E, Velázquez Cervantes MA, León Juárez M. COVID-19, a worldwide public health emergency. Rev Clin Esp. 2020;S0014-2565(20):30092–8.
WHO. Coronavirus disease (COVID-19) pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019.
Ghelichi Ghojogh M, Allah Kalteh E, Fararooei M. Coronavirus disease 2019; epidemiology and recommendations. J Prev Epidemiol. 2020;5(1):e01.
Chan JF-W, Yuan S, Kok K-H, To KK-W, Chu H, Yang J, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. The Lancet. 2020;395(10223):514–23.
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395(10223):497–506.
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9.
Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–13.
Chen L, Hao G. The role of angiotensin-converting enzyme 2 in coronaviruses/influenza viruses and cardiovascular disease. Cardiovasc Res. 2020;0:1–5.
Dadashzadeh N, Farshid S, Valizadeh R, Nanbakhsh M, Rahimi MM. Acute respiratory distress syndrome in COVID-19. Immunopathol Persa. 2020;6(2):e16.
Wang LS, Wang YR, Ye DW, Liu Q. A review of the 2019 novel coronavirus (COVID-19) based on current evidence. Int J Antimicrob Agents. 2020;55(6):105948.
Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–62.
Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, et al. Prevalence of comorbidities in the novel Wuhan coronavirus (COVID-19) infection: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91–4.
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–20.
Pearson-Stuttard J, Blundell S, Harris T, Cook DG, Critchley J. Diabetes and infection: assessing the association with glycaemic control in population-based studies. Lancet Diabetes Endocrinol. 2016;4(2):148–58.
Faghir-Gangi M, Moameri H, Abdolmohamadi N, Nematollahi SH. The prevalence of type 2 diabetes in patients with COVID-19: a systematic review and meta-analysis. Via Medica. 2020;9(5):271–8.
Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterization Protocol: prospective observational cohort study. BMJ. 2020;369:m1985.
Riddle MC, Buse JB, Franks PW, Knowler WC, Ratner RE, Selvin E, et al. COVID-19 in people with diabetes: urgently needed lessons from early reports. Diabetes Care. 2020;43(7):1378–81.
Barron E, Bakhai C, Kar P, Weaver A, Bradley D, Ismail H, et al. Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. Lancet Diabetes Endocrinol. 2020;S2213-8587(20):30272–2.
Fadini G, Morieri M, Longato E, Avogaro A. Prevalence and impact of diabetes among people infected with SARS–CoV–2. J Endocrinol Invest. 2020;43(6):867–9.
Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052–9.
Cariou B, Hadjadj S, Wargny M, Pichelin M, Al-Salameh A, Allix I, et al. Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia. 2020;63(8):1500–15.
Orioli L, Hermans MP, Thissen JP, Maiter D, Vandeleene B, Yombi JC. COVID-19 in diabetic patients: Related risks and specifics of management. Ann Endocrinol (Paris). 2020;81(2–3):101–9.
Knapp S. Diabetes and infection: is there a link? – A mini-review. Gerontology. 2013;59(2):99–104.
Petrie JR, Guzik TJ, Touyz RM. Diabetes, hypertension, and cardiovascular disease: clinical insights and vascular mechanisms. Canadian J Cardiol. 2018;34(5):575–84.
Ritz BW, Gardner EM. Malnutrition and energy restriction differentially affect viral immunity. J Nutr. 2006;136(5):1141–4.
Soares MP, Teixeira L, Moita LF. Disease tolerance and immunity in host protection against infection. Nat Rev Immunol. 2017;17(2):83.
Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6(7):438–46.
Solomons NW. Malnutrition and infection: an update. Br J Nutr. 2007;98(Suppl 1(S1):5–10.
Beck MA, Handy J, Levander OA. Host nutritional status: the neglected virulence factor. Trends Microbiol. 2004;12(9):417–23.
Acosta-Elias J, Espinosa-Tanguma R. The folate concentration and/or folic acid metabolites in plasma as factor for COVID-19 infection. Front Pharmacol. 2020;11:1062.
Narayanan N, Nair DT. Vitamin B12 may inhibit RNA-dependent-RNA polymerase activity of nsp12 from the SARS-CoV-2 virus. IUBMB Life. 2020. https://doi.org/10.1002/iub.2359.
Razdan K, Singh K, Singh D. Vitamin D, Levels. and COVID-19 Susceptibility: Is there any Correlation? Med Drug Discov. 2020;7:100051.
Reharusum P, Priambada S, Budiarti C, Agung E, Budi C. Patterns of COVID-19 mortality and vitamin D: an indonesian study. Available atSSRN 3585561. 2020. Available online: https://ultrasuninternational.com/.
Hiedra R, Bryan Lo K, Elbashabsheh M, Gul F, Wright RM, Albano J, et al. The use of IV vitamin C for patients with COVID-19: a single center observational study. Expert Rev Anti Infect Ther. 2020;5:100028.
Zhang J, Taylor EW, Bennett K, Saad R, Rayman MP. Association between regional selenium status and reported outcome of COVID-19 cases in China. Am J Clin Nutr. 2020;111(6):1297–99.
Moghaddam A, Heller RA, Sun Q, Seelig J, Cherkezov A, Seibert L, et al. Selenium deficiency is associated with mortality risk from COVID-19. Nutrients. 2020;12:2098.
Mossink JP. Zinc as nutritional intervention and prevention measure for COVID–19 disease. BMJ Nutr Prev Health. 2020;3(1):1–7.
Velthuis AJW, van den Worm SHE, Sims AC, Baric RS, Snijder EJ, van Hemer MJ. Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6(11):e1001176.
Schoen K, Horvat N, Guerreiro NFC, de Castro I, de Giassi KS. Spectrum of clinical and radiographic findings in patients with diagnosis of H1N1 and correlation with clinical severity. BMC Infect Dis. 2019;19:964.
Yang JK, Feng Y, Yuan MY, Yuan SY, Fu HJ, Wu BY, et al. Plasma glucose levels and diabetes are independent predictors for mortality and morbidity in patients with SARS. Diabet Med. 2006;23(6):623–8.
Banik GR, Alqahtani AS, Booy R, Rashid H. Risk factors for severity and mortality in patients with MERS-CoV: analysis of publicly available data from Saudi Arabia. Virol Sin. 2016;31(1):81–4.
Wang A, Zhao W, Xu Z, Gu J. Timely blood glucose management for the outbreak of 2019 novel coronavirus disease (COVID-19) is urgently needed. Diabetes Res Clin Pract. 2020;162:108118.
Varanasi SK, Donohoe D, Jaggi U, Rouse BT. Manipulating glucose metabolism during different stages of viral pathogenesis can have either detrimental or beneficial effects. J Immunol. 2017;199(5):1748–61.
Wang A, Huen SC, Luan HH, Yu S, Zhang C, Gallezot J-D, et al. Opposing effects of fasting metabolism on tissue tolerance in bacterial and viral inflammation. Cell. 2016;166(6):1512–25. e12.
O’Neill LA, Kishton RJ, Rathmell J. A guide to immunometabolism for immunologists. Nat Rev Immunol. 2016;16(9):553–65.
Stachowska E, Folwarski M, Jamioł-Milc D, Maciejewska D, Skonieczna-Żydecka K. Nutritional support in coronavirus 2019 disease. Medicina. 2020;56(6):289.
Patel JJ, Martindale RG, McClave SA. Relevant nutrition therapy in COVID-19 and the constraints on its delivery by a unique disease process. Nutr Clin Pract. 2020;35(5):792–9.
Mechanick JI, Carbone S, Dickerson RN, Hernandez BJD, Hurt RT, Irving SY, ASPEN COVID-19 Task Force on Nutrition Research. Clinical nutrition research and the COVID-19 pandemic: a scoping review of the ASPEN COVID-19 task force on nutrition research. JPEN J Parenter Enteral Nutr. 2020. https://doi.org/10.1002/jpen.2036.
Diabetes (India). National Diabetes Obesity and Cholesterol Foundation (NDOC), and Nutrition Expert Group, India. Balanced nutrition is needed. In: Times of COVID19 epidemic in India: A call for action for all nutritionists and physicians. Diabetes & Metabolic Syndrome: Clinical Research & Reviews; 2020. pp 1747–50.
Collins PF, Stratton RJ, Elia M. Nutritional support in chronic obstructive pulmonary disease: a systematic review and meta-analysis. Am J Clin Nutr. 2012;95(6):1385–95.
Chatraw JH, Wherry EJ, Ahmed R, Kapasi ZF. Diminished primary CD8 T cell response to viral infection during protein energy malnutrition in mice is due to changes in microenvironment and low numbers of viral-specific CD8 T cell precursors. J Nutr. 2008;138(4):806–12.
Ajala O, English P, Pinkney J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am J Clin Nutr. 2013;97(3):505–16.
Taylor AK, Cao W, Vora KP, Cruz JDL, Shieh W-J, Zaki SR, et al. Protein energy malnutrition decreases immunity and increases susceptibility to influenza infection in mice. J Infect Dis. 2013;207(3):501–10.
Hill K, Vogiatzis I, Burtin C. The importance of components of pulmonary rehabilitation, other than exercise training, in COPD. Eur Respir Rev. 2013;22(129):405–13.
Khan SH, Karnik AM. Medical nutrition therapy for pulmonary disease. In: Kathleen L, Mahan JLR, editors. Krause’s Food & the Nutrition Care Process. Missouri: Elsevier; 2016. p. 681–697.
Thibault R, Seguin P, Tamion F, Pichard C, Singer P. Nutrition of the COVID-19 patient in the intensive care unit (ICU): a practical guidance. Crit Care. 2020;24(1):447.
Kesteloot H. Epidemiology: past, present and future. Verh K Acad Geneeskd Belg. 2004;66(5–6):384–405.
Das UN. Polyunsaturated fatty acids and sepsis. Nutrition. 2019;65:39–43.
Husson M-O, Ley D, Portal C, Gottrand M, Hueso T, Desseyn J-L, et al. Modulation of host defence against bacterial and viral infections by omega-3 polyunsaturated fatty acids. J Infect. 2016;73(6):523–35.
Rutting S, Zakarya R, Bozier J, Xenaki D, Horvat JC, Wood LG, et al. Dietary fatty acids amplify inflammatory responses to infection through p38 MAPK signaling. Am J Respir Cell Mol Biol. 2019;60(5):554–68.
Schwerbrock NM, Karlsson EA, Shi Q, Sheridan PA, Beck MA. Fish oil-fed mice have impaired resistance to influenza infection. J Nutr. 2009;139(8):1588–94.
Rutting S, Horvat J, Wood L, Hansbro P, Oliver B. The effect of dietary fatty acids on respiratory infection in human lung cells. Eur Respir J. 2018;52:PA4983.
Kang KW, Kim S, Cho Y-B, Ryu SR, Seo Y-J, Lee S-M. Endogenous n-3 polyunsaturated fatty acids are beneficial to dampen CD8 + T cell-mediated inflammatory response upon the viral infection in mice. Int J Mol Sci. 2019;20(18):4510.
Goncalves-Mendes N, Talvas J, Duale C, Guttmann A, Corbin V, Marceau G, et al. Impact of vitamin D supplementation on influenza vaccine response and immune functions in deficient elderly persons: a randomized placebo-controlled trial. Front Immunol. 2019;10:65.
Natto ZS, Yaghmoor W, Alshaeri HK, Van Dyke TE. Omega-3 fatty acids effects on inflammatory biomarkers and lipid profiles among diabetic and cardiovascular disease patients: a systematic review and meta-analysis. Sci Rep. 2019;9(1):18867.
Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. 2020;109:102433.
Onishi JC, Häggblom MM, Shapses SA. Can dietary fatty acids affect the COVID-19 infection outcome in vulnerable populations? mBio. 2020;11(4):e01723-20.
Butler MJ, Barrientos RM. The impact of nutrition on COVID-19 susceptibility and long-term consequences. Brain Behav Immun. 2020;87:53–4.
Dushianthan A, Cusack R, Burgess VA, Grocott MP, Calder P. Immunonutrition for adults with ARDS: results from a cochrane systematic review and meta-analysis. Respir Care. 2020;65(1):99–110.
Langlois PL, D’Aragon F, Hardy G, Manzanares W. Omega-3 polyunsaturated fatty acids in critically ill patients with acute respiratory distress syndrome: A systematic review and meta-analysis. Nutrition. 2019;61:84–92.
Turner KL, Moore FA, Martindale R. Nutrition support for the acute lung injury/adult respiratory distress syndrome patient: a review. Nutr Clin Pract. 2011;26(1):14–25.
Maggini S, Wintergerst ES, Beveridge S, Hornig DH. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br J Nutr. 2007;98:29–35.
Saeed F, Nadeem M, Ahmed RS, Tahir Nadeem M, Arshad MS, Ullah A. Studying the impact of nutritional immunology underlying the modulation of immune responses by nutritional compounds – a review. Food Agric Immunol. 2016;27(2):205–29.
Bunout D, Barrera G, Hirsch S, Gattas V, de la Maza MP, Haschke F, et al. Effects of a nutritional supplement on the immune response and cytokine production in free-living Chilean elderly. J Parenter Enteral Nutr. 2004;28(5):348–54.
Smith AD. Folic acid fortification: the good, the bad, and the puzzle of vitamin B-12. Am J Clin Nutr. 2007;85:3–5.
Duthie SJ, Horgan G, De Roos B, Rucklidge G, Reid M, Duncan G, et al. Blood folate status and expression of proteins involved in immune function, inflammation, and coagulation: biochemical and proteomic changes in the plasma of humans in response to long-term synthetic folic acid supplementation. J Proteome Res. 2010;9(4):1941–50.
Al-Maskari MY, Waly MI, Ali A, Al-Shuaibi YS, Ouhtit A. Folate and vitamin B12 deficiency and hyperhomocysteinemia promote oxidative stress in adult type 2 diabetes. Nutrition. 2012;28(7–8):e23–6.
Erkurt MA, Aydogdu I, Dikilitaş M, Kuku I, Kaya E, Bayraktar N, et al. Effects of cyanocobalamin on immunity in patients with pernicious anemia. Med Princ Pract. 2008;17:131–5.
Kim J, Woo Ahn C, Fang S, Lee H, Suk Park J. Association between metformin dose and vitamin B12 deficiency in patients with type 2 diabetes. Medicine. 2019;98(46):e17918.
Wakeman M, Archer T. Metformin and micronutrient status in type 2 diabetes: does polypharmacy involving acid-suppressing medications affect vitamin B12 levels? Diabetes Metab Syndr Obes. 2020;13:2093–108.
Aroda VR, Edelstein SL, Goldberg RB, Knowler WC, Marcovina SM, Orchard TJ, et al. Long-term metformin use and vitamin B12 Deficiency in the diabetes prevention program outcomes study. J Clin Endocrinol Metab. 2016;101(4):1754–61.
Jovic TH, Ali SR, Ibrahim N, Jessop ZM, Tarassoli SP, Dobbs TD, et al. Could vitamins help in the fight against COVID-19? Nutrients. 2020;12(9):E2550.
Zhang R, Wang X, Ni L, Di X, Ma B, Niu S, et al. COVID-19: Melatonin as a potential adjuvant treatment. Life Sci. 2020;250:117583.
Yanagihara M, Nakamura M, Usui A, Nishida S, Ito E, Okawa M, et al. The melatonin receptor agonist is effective for free-running type circadian rhythm sleep disorder: case report on two sighted patients. Tohoku J Exp Med. 2014;234(2):123–8.
dos Santos LMJ. Can vitamin B12 be an adjuvant to COVID-19 treatment? GSC Biol Pharm Sci. 2020;11(03):001–5.
Coutard B, Valle C, de Lamballerie X, Canard B, Seidah NG, Decroly E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral Res. 2020;176:1872–90.
Wu C, Zheng M, Yang Y, Gu X, Yang K, Li M, et al. Furin, a potential therapeutic target for covid-19. iScience. 2020;23(10):101642.
Sheybani Z, Dokoohaki MH, Negahdaripour M, Dehdashti M, Zolghadr H, Moghadami M, et al. The role of folic acid in the management of respiratory disease caused by covid-19. Available online: https://chemrxiv.org/articles/preprint/The_Role_of_Folic_Acid_in_the_Management_of_Respiratory_Disease_Caused_by_COVID-19/12034980/1.
Rosen CJ, Adams JS, Bikle DD, Black DM, Demay MB, Manson JE, et al. The nonskeletal effects of vitamin D: an Endocrine Society scientific statement. Endocr Rev. 2012;33(3):456–92.
Bland R, Markovic D, Hills CE, Hughes SV, Chan SL, Squires PE, et al. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in pancreatic islets. J Steroid Biochem Mol Biol. 2004;89–90:121–5.
Zehnder D, Bland R, Williams MC, McNinch RW, Howie AJ, Stewart PM, et al. Extrarenal expression of 25-hydroxyvitamin d(3)-1 alpha-hydroxylase. J Clin Endocrinol Metab. 2001;86(2):888–94.
Gysemans CA, Cardozo AK, Callewaert H, Giulietti A, Hulshagen L, Bouillon R, et al. 1,25-Dihydroxyvitamin D3 modulates expression of chemokines and cytokines in pancreatic islets: implications for prevention of diabetes in nonobese diabetic mice. Endocrin. 2005;146:1956–64.
Park S, Kim DS, Kang S. Vitamin D deficiency impairs glucose-stimulated insulin secretion and increases insulin resistance by reducing PPAR-gamma expression in nonobese Type 2 diabetic rats. J Nutr Biochem. 2016;27:257–65.
Zhou QG, Hou FF, Guo ZJ, Liang M, Wang GB, Zhang X. 1,25-Dihydroxyvitamin D improved the free fatty-acid-induced insulin resistance in cultured C2C12 cells. Diabetes Metab Res Rev. 2008;24:459–64.
Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients. 2015;7(6):4240–70.
Zisi D, Challa A, Makis A. The association between vitamin D status and infectious diseases of the respiratory system in infancy and childhood. Hormones (Athens) 2019;18(4):353–63.
Teymoori-Rad M, Shokri F, Salimi V, Marashi SM. The interplay between vitamin D and viral infections. Rev Med Virol. 2019;29(2):e2032.
Zdrenghea MT, Makrinioti H, Bagacean C, Bush A, Johnston SL, Stanciu LA. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev Med Virol. 2017;27(1):e1909.
Lee MD, Lin CH, Lei WT, Chang HY, Lee HC, Yeung CY, et al. Does vitamin D deficiency affect the immunogenic responses to influenza vaccination? A systematic review and meta-analysis. Nutrients. 2018;10(4):409.
Kohlmeier M. Avoidance of vitamin D deficiency to slow the COVID-19 pandemic. BMJ Nutr Prev Health. 2020;3(1):1–7.
Grant WB, Lahore H, McDonnell SL, Baggerly CA, French CB, Aliano JL, et al. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients. 2020;12(4):988.
Nurshad A. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J Infect Public Health. 2020;1390:8.
Baeke F, Takiishi T, Korf H, Gysemans C, Mathieu C. Vitamin D: modulator of the immune system. Curr Opin Pharmacol. 2010;10(4):482–96.
PanareseA SE. Covid-19, and vitamin D. Aliment Pharmacol Ther. 2020;51(10):993–5.
Ebadi M. Montano-Loza AJ. Perspective: improving vitamin D status in the management of COVID-19. Eur J Clin Nutr. 2020;74:856–9.
Merzon E, Tworowski D, Gorohovski A, Vinker Sh, Cohen AG, Green I, et al. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population based study. FEBS J. 2020;287(17):3693–02.
Ginde AA, Blatchford P, Breese K, Zarrabi L, Linnebur S, Wallace J, et al. High-dose monthly vitamin D for prevention of acute respiratory infection in older long-term care residents: a randomized clinical trial. J Am Geriatr Soc. 2017;65(3):496–503.
Young JI, Züchner S, Wang G. Regulation of the epigenome by vitamin C. Annu Rev Nutr. 2015;35:545–64.
Castro SM, Guerrero-Plata A, Suarez-Real G, Adegboyega PA, Colasurdo GN, Khan AM, et al. Antioxidant treatment ameliorates respiratory syncytial virus–induced disease and lung inflammation. Am J Respir Crit. 2006;174(12):1361–9.
Hemilä H, Chalker E. Vitamin C can shorten the length of stay in the ICU: a meta analysis. Nutrients. 2019;11:708.
Gonzalez M, Berdiel M, Duconge J, Levy Th, Alfaro I, Morales-Borges R, et al. High dose vitamin C and influenza: a case report. J Orthomol Med. 2018;33:1–3.
Ran L, Zhao W, Wang J, Wang H, Zhao Y, Tseng Y, et al. Extra dose of vitamin C based on a daily supplementation shortens the common cold: A meta-analysis of 9 randomized controlled trials. Biomed Res Int. 2018;1–12.
Zhang J, Wang X, Vikash V, Ye Q, Wu D, Liu Y, et al. ROS and ROS-mediated cellular signaling. Oxid Med Cell Long. 2016;4350965:1–18.
Praveen D, Puvvada RC, Aanandhi VA. Association of vitamin C status in diabetes mellitus: prevalence and predictors of vitamin C deficiency. Future J Pharm Sci. 2020;6:30.
Das UN. Vitamin C for type 2 diabetes mellitus and hypertension. Arch Med Res. 2019;50(2):11–4.
Varma V, Varma M, Dey Sarkar P, Varma A, Vyas S, Kulkarni R. Correlation of vitamin C with HbA1c and oxidative stress in diabetes mellitus with or without nephropathy. Natl J Med Res. 2014;4(2):151–5.
Cheng R. Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)? Med Drug Discov. 2020;5:100028.
Rayman MP. Selenium and human health. Lancet. 2012;379:1256–68.
Ng MP, Lee JC, Loke WM, Yeo LL, Quek AM, Lim EC, et al. Does influenza A infection increase oxidative damage? Antioxid Redox Signal. 2014;21(7):1025–31.
Erkekoğlu P, Aşçı A, Ceyhan M, Kızılgün M, Schweizer U, Ataş C, et al. Selenium levels, selenoenzyme activities and oxidant/antioxidant parameters in H1N1-infected children. Turk J Pediatr. 2013;55(3):271–82
Wei J, Zeng Ch, Gong Q, Yang H, Li X, Lei G, Yang T. The association between dietary selenium intake and diabetes: a cross-sectional study among middle-aged and older adults. Nutr J. 2015;14:18.
Kljai K, Runje R. Selenium and glycogen levels in diabetic patients. Biol Trace Elem Res. 2001;83:223–9.
Hiffler L, Rakotoambinina B. Selenium and RNA virus interactions: Potential implications for SARS-CoV-2 infection (COVID-19). Front Nutr. 2020;7:164.
Jinsong Zhang J, Taylor E, Bennett K, Saad R, Rayman M. Selenium supplementation in the prevention of coronavirus infections (COVID-19). Med Hypotheses. 2020;143:109878.
Maares M, Haase H. Zinc and immunity: An essential interrelation. Arch Biochem Biophys. 2016;611:58–65.
Clímaco Cruz KJ, Oliveira A, Marreiro D. Antioxidant role of zinc in diabetes mellitus. World J Diabetes. 2015;15(6(2):333–7.
Miao X, Sun W, Fu Y, Miao L, Cai L. Zinc homeostasis in the metabolic syndrome and diabetes. Front Med. 2013;7:31–52.
Capdor J, Foster M, Petocz P, Samman S. Zinc and glycemic control: a meta-analysis of randomised placebo controlled supplementation trials in humans. J Trace Elem Med Biol. 2013;27:137–42.
Poliana Guiomar de Almeida Brasiel. The key role of zinc in elderly immunity: A possible approach in the COVID-19 crisis. Clin Nutr. 2020;38:65–6.
Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14:353–7.
Carocho M, Ferreira IC. A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem Toxicol. 2013;51:15–25.
Han YS, Chang GG, Juo CG, Lee HJ, Yeh SH, Hsu JT, et al. Papain-Like protease 2 (PLP2) from severe acute respiratory syndrome coronavirus (SARS-CoV): expression, purification, characterization, and inhibition. Biochemistry. 2005;44:10349–59.
Shittu MO, Afolami OI. Improving the efficacy of Chloroquine and Hydroxychloroquine against SARS-CoV-2 may require Zinc additives - A better synergy for future COVID-19 clinical trials. Infez Med. 2020;28:192–7.
Speth R, Carrera E, Jean–Baptiste M, Joachim A, Linares A. Concentration–dependent effects of zinc on angiotensin–converting enzyme–2 activity. FASEB J. 2014;28(S1):1067.4.
Arentz S, Hunter J, Yang G, Beardsley J, Myers SP, Mertz D, et al. Zinc for the prevention and treatment of SARS-CoV-2 and other acute viral respiratory infections: a rapid review. Adv Integr Med. 2020;7:252–60.
Acknowledgements
We wish to express our appreciation to the Research Vice-Chancellor of Tabriz and Maragheh Universities of Medical Sciences.
Author information
Authors and Affiliations
Contributions
SM, VEA, MSA contributed in designing the study, searching for resources and writing the manuscript. JH and MJ cooperated in completing the mechanism of action for nutrients. JRH contributed in scientific revising of manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
It is not applicable.
Consent for publication
It is not applicable.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahluji, S., Jalili, M., Ostadrahimi, A. et al. Nutritional management of diabetes mellitus during the pandemic of COVID-19: a comprehensive narrative review. J Diabetes Metab Disord 20, 963–972 (2021). https://doi.org/10.1007/s40200-021-00784-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00784-5